DOH: Only 21 out of 114,000 COVID-19 vaccine recipients had serious side effects
MANILA, Philippines — Twenty-one serious side effects after vaccinations against COVID-19 have been so far reported, the Department of Health said Friday.
Out of the 114,615 people who have been inoculated as of March 10, 978 or only 0.85% experienced suspected adverse events after immunization (AEFI), Health Undersecretary Maria Rosario Vergeire.
AEFI “is any untoward medical occurrence which follows immunization and which does not necessarily have a causal relationship with the usage of the vaccine,” according to the World Health Organization.
For those vaccinated with Coronavac, or the vaccine developed by China’s Sinovac Biotech, 872 experienced minor side effects such as pain on the injection site, body pains, increase in blood pressure, fever and rashes.
Eighty-five recipients of the vaccine made by British-Swedish firm AstraZeneca also had common and non-serious side effects.
Meanwhile, 20 CoronaVac recipients and one AstraZeneca recipient experienced serious side effects such as difficulty in breathing and chest pain.
“We’re studying the causality,” Vergeire said.
The health official said the National Adverse Events Following Immunization Committee believes that some of the cases can be attributed to “anxiety due to fear of side effects or needles.”
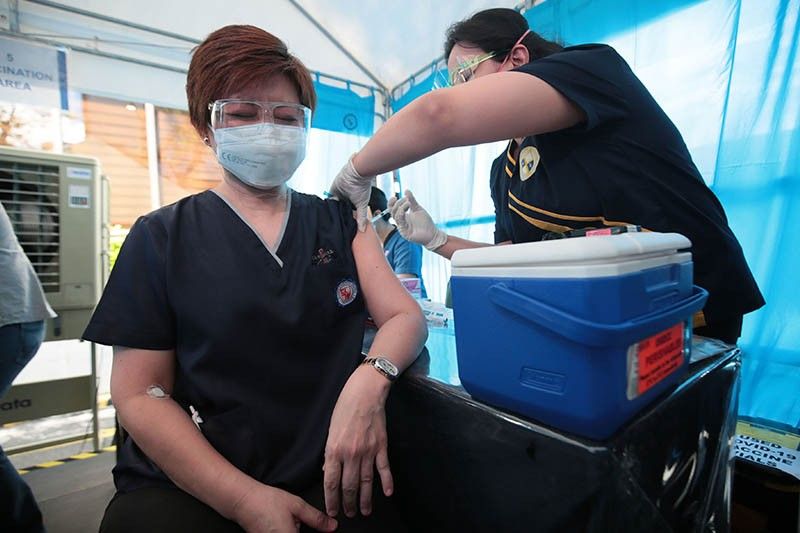
After several delays, the government began its vaccination program against COVID-19 last week, with health workers and uniformed personnel first in the queue to get donated shots. Some 101,827 people received CoronaVac, while 12,788 got the AstraZeneca jab.
The department has a process for monitoring adverse events after immunization. Those who received the shots are made to stay in the vaccination site for half an hour in case of immediate reaction.
The country's Food and Drug Administration, in the emergency use authorizations it has issued, said the benefits of approved vaccines outweigh potential risks. Only the COVID-19 vaccine of Pfizer-BioNTech, AstraZeneca and Sinovac have so far received EUA from the drug regulator.
- Latest
- Trending