FDA: Sinopharm vaccine given to Duterte covered by special permit
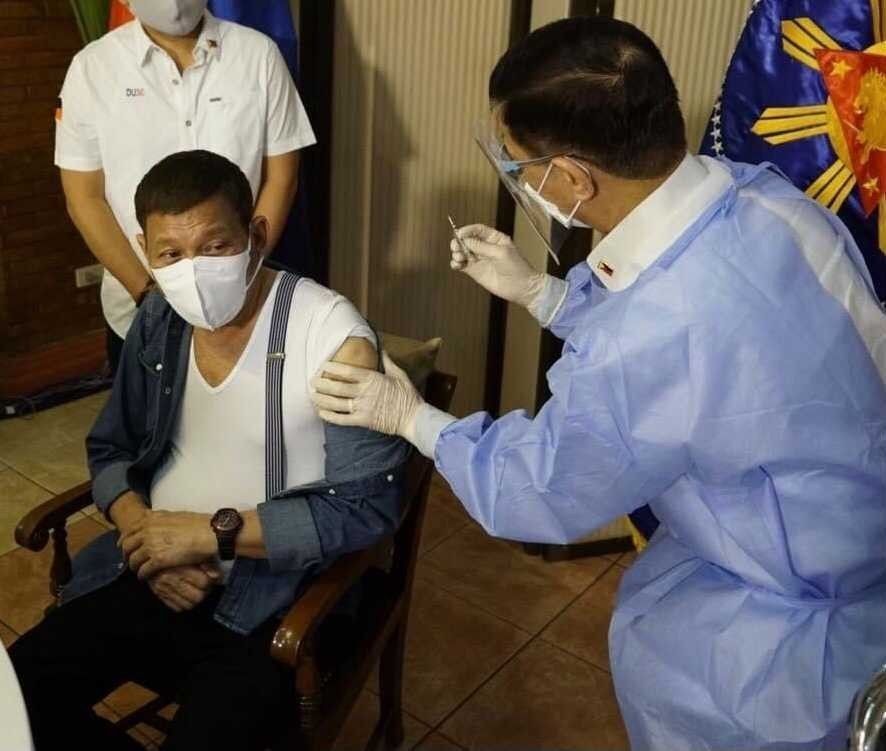
MANILA, Philippines — The country’s Food and Drug Administration said the COVID-19 vaccine administered to President Rodrigo Duterte was covered by the compassionate use permit granted to the chief executive’s security group.
FDA Director General Eric Domingo said this in an interview with ABS-CBN’s Teleradyo Tuesday after Duterte received his first shot of the jab developed by Sinopharm Group Co. Ltd.
The country’s FDA has yet to approve the vaccine for emergency use. The Chinese-made jab has been restricted to compassionate use only in the Philippines.
In February, the FDA granted the Presidential Security Group’s application for compassionate special permit to access 10,000 doses of the Sinopharm vaccine. A CSP allows the use of unregistered medical products, such as the Sinopharm jab, for limited off-label use.
“When you say compassionate special permit, it’s not an authorization given by the FDA. In this case, the head of the PSG hospital guarantees they studied the vaccine and they take full responsibility for it,” Domingo said.
It is unclear why Duterte, a president, was covered by the permit given to his security team. But Domingo said the wives and partners of PSG personnel are also covered by the CSP.
The vaccine given to Duterte was the same one illegally administered to his security group before any jab had been approved for emergency use.
FDA yet to study Sinopharm jab
The FDA chief also said his agency has yet to evaluate the vaccine injected to Duterte by Health Secretary Francisco Duque III.
“The FDA has yet to study it (Sinopharm vaccine),” Domingo said.
He said there were EUA applications for the Sinopharm vaccines but parties have yet to submit requirements.
The chief executive has previously expressed confidence in vaccines made in China and Russia.
Currently, the vaccines developed by Sinovac, AstraZeneca and Gamaleya Research Institute are being rolled out in the Philippines.
Since the start of the vaccination program in March, over 1.6 million people have received first dose, while fewer than 300,000 have been fully vaccinated.
The national government has so far secured two official deals for COVID-19 vaccine supplies in the Philippines, one with Chinese pharmaceutical company Sinovac and another with the Serum Institute of India.
Watch this space for bite-sized developments on the vaccines in the Philippines. (Main image by Markus Spiske via Unsplash)
Health Officer-in-Charge Maria Rosario Vergeire says the general population may now get their second booster jab.
"We're just waiting for the release of implementing guidelines, then we'll start rolling out our second booster for the general population," she says. — Gaea Katreena Cabico
Amid questions on vaccines being administered, the Department of Health assures the public all doses are safe and effective as the “process of extending shelf life goes through thorough stability studies.”
“The government ensures that every vaccine that is injected with an extended shelf life has gone through studies, and is still safe and effective against COVID-19,” it adds.
Government must increase vaccination capacity across the Philippines in anticipation of a surge of COVID-19 cases caused by the Omicron variant of the corona virus, Sen. Risa Hontiveros says.
She says local government units and the private sector can work together to put up more vaccination centers and deploy more vaccination teams to get more people inoculated against COVID-19.
"The active COVID cases have nearly doubled in three days. The positivity rate is almost four times the ceiling set by the World Health Organization. Huwag na nating hintayin na sobrang lumala pa ang sitwasyon bago tayo gumawa ng paraan para mapabilis ang ating pagbabakuna."
FDA chief Eric Domingo says that its agency has given emergency approval for the use of the Pfizer-BioNTech COVID-19 vaccine for children ages 5 to 11.
The United States immunized around 900,000 children aged five-to-11 against Covid in the first week the Pfizer vaccine was authorized for them, a White House official says Wednesday.
Roughly 700,000 more have made appointments at pharmacies, White House Covid coordinator Jeff Zients tells reporters.
"The program is just getting up to full strength," he says, adding most of the shots were given in the last couple of days alone. — AFP
- Latest
- Trending