Breakthrough drug that cured Jimmy Carter of cancer now in the Philippines
On August 3, 2015, former US President Jimmy Carter, peanut farmer from Georgia, rabid social activist, and Nobel Peace Prize awardee, was wheeled in for surgery to remove a small mass from his liver. Nine days later, he disclosed that he had been diagnosed with liver cancer that had spread to other parts of his body. (Carter has an extensive family history of cancer — both his parents and all of his three siblings had cancer.) Eight days later, he revealed in a news conference that doctors had found melanoma — “four very small spots” — on his brain and that he had began treatment with the immunotherapy drug Pembrolizumab and would start radiation soon. Like a scarred soldier marching off to another war, Carter bravely faced another bruising battle with the Big C as he announced, “I’m perfectly at ease with whatever comes. I have led a wonderful life. Now, I feel it’s in the hands of God.”
On Dec. 6, 2015, he declared he was cancer-free.
Thus, news about this wonder drug called Pembrolizumab that cured Jimmy Carter spread like wildfire. And now, that drug is available in the Philippines. In February this year, the Food and Drug Administration of the Philippines approved Pembrolizumab for the treatment of advanced or metastatic lung cancer and melanoma, a type of skin cancer.
Dr. Ena Lyn Ang, medical adviser, MSD Oncology, notes that in the Philippines, lung cancer is still the top cause of mortality among men and it’s top two if you combine men and women. She says that majority of the patients are diagnosed at a late stage — or Stage IIIB, which is inoperable, and Stage IV or metastatic, where the tumor has spread to distant parts of the body.
On the other hand, melanoma accounts for less than five percent of all types of skin cancer but is the most difficult to treat and responsible for the most skin cancer deaths as it can spread to other parts of the body.
Now comes Pembrolizumab, a bright ray of hope for hopeless cases.
“Addressing the rising incidence of cancer is a tough road to embark on. MSD has been dedicating its resources to develop innovative oncology medicines. Pembrolizumab propelled our research efforts to understand the role of the immune system and the PD-L1 pathway in cancer treatment,” said Dr. Beaver Tamesis, president and managing director, MSD Philippines, the pharmaceutical company behind Pembrolizumab.
Did you know that May is Melanoma Awareness Month? Here are excerpts from the Q&A at the press conference hosted by MSD:
What is immunotherapy and how does it work?
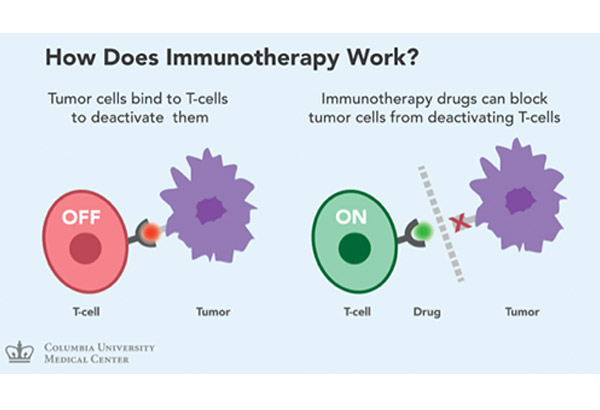 Immunotherapy activates the immune system to fight cancer.
Immunotherapy activates the immune system to fight cancer.DR. GERARDO CORNELIO (head, Cancer Institute, St. Luke’s Medical Center Global City): Immunotherapy activates the immune system to fight cancer. I think last year, it was awarded by the American Society of Clinical Oncology as the breakthrough agent in oncology. It uses certain parts of the immune system to fight diseases such as cancer by stimulating your own system to work harder or smarter to attack cancer cells. It gives one’s immune system components such as man-made immune system proteins. There are different types of immunotherapy — there are vaccines, enhancement of antigen presentation and cytokines. These are the older version of immunotherapy drugs, which really did not show much promise before.
Tyrosine Kinase Inhibitors are targeted therapies which use biomarkers — these are directed against cancer-specific molecules, and signaling pathways, and have more limited non-specific toxicities. The drugs that are approved right now on the market would be Erlonitib, Gefitinib, and Afatinib. Crizotinib is a different kind of drug and belongs to a different family. The limitations would be development of resistance, although now, we have also been able to detect which specific resistance these drugs have developed, and there are also new third-generation Tyrosine Kinase Inhibitors that are coming up.
Tumor cells bind to T-cells to deactivate these cells. If you look at these specific cells, this is a T-cell, this is a tumor cell, this is deactivated so it does not really do anything. When immunotherapy drugs block the tumor cells from deactivating the T-cells, these T-cells are activated, thereby attacking the tumor cell. So, it makes your own immune system recognize the tumor cells.
In layman’s terms, so if you’re probably familiar with science fiction movies, if you watched Independence Day, you have the alien starship, and there’s a force field and then you have these rebels that are trying to attack this alien ship. Without the force field, then this rebel ship attacking the alien ship would be able to penetrate and make it disintegrate. So, this is the same way that these tumor cells work. This is how immune checkpoint inhibitors work.
What is a biomarker?
A biomarker is a biological molecule that is found in the blood or body fluids, or tissues, and is a sign of normal or abnormal processes or a condition of disease. These biomarkers may be used to see how well the body responds to a treatment or a disease condition.
The biomarker testing is a standard of care now in advanced non-small cell lung cancer, specifically for adenocarcinoma. This allows us to choose which drugs we will have to give to our patients.
The emerging biomarker used for new-generation immunotherapy is PD-L1, so we are really waiting for this, and hopefully, MSD will be able to help us get this biomarker available for our patients.
Moving now to Pembrolizumab, this was published a few years ago in the New England Journal of Medicine, “Pembrolizumab for the treatment of non-small cell lung cancer.”
The NCCN (National Comprehensive Cancer Network) Guidelines, which is a group of top cancer centers across the United States, has now included Pembrolizumab as a preferred regimen in first-line metastatic, advanced melanomas, together with Ipilimumab, Nivolumab, and of course, putting patients in clinical trials.
We are now in the process of also looking at the benefit of these drugs across all cancer types (such as head and neck cancers, urethral cancer, gastric malignancies, triple negative breast cancer, Hodgkin’s Lymphoma). I’m actually one of the investigators of this trial.
How is Pembrolizumab administered?
DR. CORNELIO: Pembrolizumab is given intravenously. We use 2mg/kg, but in clinical trials, they use 2 and 10mg/kg. Now, the benefit is very much the same. Of course, we try to use the lower one if the benefit is the same. It’s given intravenously every three weeks, 30 minutes, so it’s very easy to give, with of course less side effects compared to cytotoxics.
What are some of the side effects that a patient might experience?
DR. CORNELIO: With the use of cytotoxic chemotherapy, since it’s cytotoxic, it’s toxic to the cells, although as oncologists, we were able to determine how to treat these side effects. These patients still undergo a process such as hair loss, vomiting, the risk of infection, because their white cell counts may drop. On the other hand, Pembrolizumab more or less does not really affect these cells and therefore the side effects are more of what we call immunologic. When you say immunologic, we prepare these patients before we give them, we check their thyroid function test, their Hepatitis B profile, liver function test, kidney function test, because the side effects are more thyroiditis, colitis, and so on. It’s more of inflammation. It may happen or it may not happen, or it may happen several months after the treatment, so we just have to monitor the patient.
DR. ENA LYN ANG: Based on the clinical trials of Pembrolizumab, these potential immune-related adverse events, the intensity, the severity, the high grade would be mostly less than one percent. As Dr. Cornelio mentioned, the most common is hypothyroidism, but based on investigators, as long as they give the appropriate treatment to control the hypothyroidism, you can actually continue giving the drug.
How much does Pembrolizumab cost?
ALLAN ANCHETA (business unit director for diversified brands, hospital and specialty care, MSD Philippines): As with any innovative cancer therapy, there is a range of price for Pembrolizumab, the price is about a hundred thousand pesos up and it will vary again based on the condition of the patient and the dosing recommended by the medical oncologist. We at MSD in the Philippines make sure that the best of our medicines are made available to the broad number of patients as much as possible.
For cancer patients who cannot afford the treatment, what help or other options are available?
ANCHETA: Right now, MSD in the Philippines is currently under discussions with several stakeholders, government, private institutions, even insurance companies to make sure that this drug is made widely available.
How does it affect treatment if a person has an immunological disease?
DR. CORNELIO: We have not really tried giving it together with other agents, but there are trials that are trying to combine it with chemotherapy, so we do not know yet how beneficial, or is there any drug-to-drug interaction with these agents, if they’re combined with another drug. We’re still in that stage where we’re using it as a single agent for non-small cell lung cancer, for melanomas, and all the tumor types that have been approved. With regard to those patients already with an autoimmune disease, we just request for clearance. If these patients have a co-existing thyroid problem, I refer them to an endocrinologist and have the disease controlled, and we just monitor the patients while on the treatment.
DR. ANG: On auto-immunity, as of now, we don’t have data to support the use of immunotherapy on these patients. In fact, on the clinical trials being done on Pembrolizumab, it’s actually one of the exclusion criteria, so patients with auto-immune problems.
Are there any biomarker testing centers here in the Philippines?
DR. ANG: In the Philippines, we have identified six centers that have the machine to test the biomarker PD-L1. In Metro Manila, we have four — that’s Asian Hospital, PGH, Lung Center, and NKTI — and two in Cebu. So, as for how patients can get tested, we’ll give you more information on that later on.
Why do some patients drop out of treatment?
DR. CORNELIO: Based on experience, patients drop out because they already feel good, or because of the cost.
Patients like to take supplements because these strengthen the immune system, will it work with immunotherapy?
DR. CORNELIO: I don’t stop my patients from taking supplements as long as it is not too much. I would like to mention a study mentioned to us about four years ago; there was a trial that was presented to us at the American Society of Clinical Oncology Conference where they randomized lung cancer patients between chemotherapy plus those patients with a high dose of antioxidants using selenium, while another arm was given chemotherapy alone. That was a big, big trial and in that study, there were more deaths seen in the group of patients with a high dose of antioxidants with chemotherapy arm. So, we were all surprised and we started moving away or discouraging patients from taking a high dose of antioxidants because of that. What we do not know is whether these drugs will actually interfere with the treatment, or it may actually boost tumor cells, making it more difficult for radiation therapy or chemotherapy to work.
Is there a guarantee that the lung cancer or melanoma would be prevented from coming back if the patient receives immunotherapy?
DR. CORNELIO: I don’t think there is a study yet on immunotherapy, unless there are studies ongoing in the early stage, what we call adjuvant therapy. If you notice when we treat patients, specifically breast cancer, for example, after surgery, we offer them systemic chemotherapy to prevent recurrence of the disease. Now, with immunotherapy, I don’t know if there are studies yet, there’s none yet, but I’m sure there will be probably in the future using this in the early stages.
Can immunotherapy be a preventive measure, too?
DR. ANG: For now, all the studies are directed towards late-stage cancer so nothing on early stage.
Others opt not to get treatment because of their age, how do see immunotherapy helping the elderly?
DR. CORNELIO: I’ve used this drug in many of my patients already. The first patient I’ve used this on is a 76 or 77-year-old patient on a very bad performance status or functional status, a bed-ridden patient, in the ICU with recurrence of the non-small cell lung cancer in both lungs with water. She had a tube in the chest, we had several lines of chemotherapy given to her, including radiation therapy for more than two to three years. So, this patient could not be given chemotherapy anymore, definitely, we discussed with the family about this drug, immunotherapy, and they were willing to explore it. We tried and this patient responded very well and we were able to take out the tube. In short, I think in terms of toxicities, we would be more comfortable giving this to elderly patients compared to chemotherapy although now, there are ways to give chemotherapy even to elder patients, but then again, you choose your patients.
DR. CESAR RECTO II (medical affairs director, MSD Philippines): We had a pre-licensed supply prior to the approval of our drug and the eldest patient we have is more than 90 years old, and she’s still doing very well after several cycles of the drug.
The drug is given intravenously. Will it be out-patient or in-patient? And how long does the patient need to stay in the treatment room?
DR. CONELIO: I give it as out-patient and you need to stay in the treatment room for 30 minutes. Actually, there are no pre-medications, but I give a little bit of pre-medications just for my peace of mind. I try to get all the thyroid function test, the liver function test, and the clearances that I need.
After 30 minutes, patients can walk out of the clinic, possibly go to a bar, drink and be merry, right?
DR. CORNELIO: Yes. But I don’t want to keep everybody’s hopes so high, this is not a wonder drug that works for everybody; there are specific tumors that it will work on and there are tumors where it may not work.
* * *
For inquiries on the drug that cured Jimmy Carter, call 784-9500, fax 885-0773 to 75, email inquiries.philippines@merck.com.



















